Abstract
Background
During 2020, the novel COVID-19 pandemic lead to cryopreservation of allogeneic hematopoietic stem cell (HSCT) grafts based on NMDP and EBMT recommendations, to secure grafts before start of conditioning chemotherapy. We examined the impact of this change in practice on patient outcomes.
Methods
We retrospectively analyzed the outcomes of 483 patients who received HSCT between Aug 2017 and Aug 2020, at Princess Margaret Cancer Centre, Canada, comparing the outcomes between those who received cryopreserved (CRYO, n=135) or fresh peripheral blood stem cell grafts (FRESH, n=348). Median follow-up: 12.3 months.
Probability of overall survival (OS) was calculated using the Kaplan-Meier product-limit method and heterogeneity of time-to-event distribution functions were compared by the log-rank test. Cumulative incidences of aGvHD and cGvHD, relapse, and non-relapse mortality (NRM) were estimated using the cumulative incidence method considering competing risk, and groups were compared using Gray's test. Death was considered as a competing event for relapse, aGvHD and cGvHD, and relapse was considered a competing event for NRM, aGvHD and cGvHD.
Results
Median age was 58y; 54.5% were males. Acute myeloid leukemia was commonest HSCT indication (n=248, 49.1%). Donors: MUD 10/10 n=233; MUD 9/10 (MMUD) n=48, matched related donor (MRD) n=112, Haploidentical n=88. Transplant conditioning: 79 (23%) and 23 (17%) patients received myeloablative conditioning (MAC) in the FRESH and the CRYO groups, respectively (p ns). In the FRESH group, 253 (73%) patients and 114 (84%) patients in the CRYO group received ATG followed by posttransplant cyclophosphamide (PTCy) and Cylosporine GvHD prophylaxis.
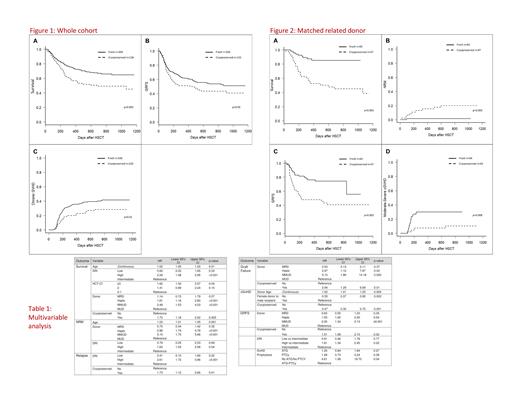
OS at the 2y timepoint, FRESH group (n=348), was 67.0% (61.1-72.3%), compared to 48.7% (38.1-58.4%) for patients in the CRYO group (n=135), p=0.002, Figure 1a. This was mainly due to MRD cohort outcomes: 2y OS in MRD FRESH group (n=65), was 85.2% (73.3-92.0%), compared to 45.1% (29.9-59.1%) in MRD CRYO group (n=47), p<0.001, Figure 2a. Multivariate analysis (MVA) for OS, significant factors were increasing patient age, DRI high/v.-high, HCT-CI ≥ 3, Donors: Haplo and MMUD, cryopreservation, Table 1.
NRM at 1y for FRESH (n=348) 17% (13.2-21.2) vs CRYO (n=135) 22.1 % (14.8-30.4), p ns . However, in the MRD cohort, NRM at 1y for FRESH group (n=65), was 1.5% (0.1-7.4%), compared to 15.4% (6.6-27.4%) for CRYO group (n=47), p=0.003, Figure 2b . On MVA, NRM adverse significant factors were patient age, DRI high/v.-high, Donors: Haplo and MMUD, Table 1.
Cumulative incidence (CI) of relapse at 2y for FRESH 22.4% (17.5-27.7) vs CRYO 27.0 % (18.8-35.9) p=0.07 .
The CI of moderate-severe cGvHD at 1y for FRESH group (n=315) was 21.5% and 10.8% in the CRYO group, p=0.027, Figure 1c . Patients with FRESH 10/10 MUDs (n=180), had CI of moderate-severe cGvHD at 1y of 20.6%, compared to CRYO 10/10 MUDs (n=35), 6.0% p ns for MUDs; FRESH MRD (n=64) CI was 30.1%; and CRYO MRD (n=43) 10.3%, p=0.008 for MRD, Figure 1d. On MVA, significant adverse factors for chronic GvHD were increasing donor age, male recipient/female donor, whilst graft CRYO was protective, Table 1.
GvHD-and Relapse free Survival (GRFS) at 2y for FRESH 54.0% (47.9-59.6) vs CRYO 43.4% (33.4-53.0) p<0.05, Figure 1b. However, in the MRD cohort, GRFS at 2y in FRESH group (n=65), was 74.2% (61.3-83.4%), compared to 40.7% (26.3-54.6%) for CRYO group (n=47), p=0.001; other donor types no difference, Figure 1c. On MVA, significant factors correlated with worse GRFS were: DRI high and very-high, cryopreservation, Donors: Haplo and MMUD, Table 1.
Compared to FRESH group, CRYO group experienced reduced cGvHD, delay in neutrophil engraftment, higher graft failure and increased CMV reactivation, with no difference in relapse incidence or acute GvHD.
Conclusion
Cryopreservation was associated with inferior outcomes post-HSCT particularly in the MRD cohort, possibly due to combination ATG and post-transplant cyclophosphamide impacting differential tolerance to cryopreservation on components of the stem cell graft; further studies are warranted to elucidate mechanisms for this observation.
Law: Novartis: Consultancy; Actinium Pharmaceuticals: Research Funding. Lipton: Bristol Myers Squibb, Ariad, Pfizer, Novartis: Consultancy, Research Funding. Kim: Bristol-Meier Squibb: Research Funding; Paladin: Honoraria, Research Funding; Pfizer: Honoraria, Research Funding; Novartis: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding. Mattsson: MattssonAB medical: Current Employment, Current holder of individual stocks in a privately-held company.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal